Choosing a Specification
Why a Breathing Air Specification May Not Be the Right Choice for Manufacturers.
Compressed Air Testing
Compressed air is a critical utility used in over 90% of manufacturing facilities. Compressed air used for industrial purposes is in a different category from compressed air used for medical and breathing purposes. You would think that limits for breathing air would be more stringent than manufacturing air – but that’s not the case.
Breathing air specifications are primarily concerned with gases like carbon monoxide (the silent killer), carbon dioxide, oxygen percentage, total gaseous hydrocarbons, water in some cases, and condensed hydrocarbons (oil mist). The limits for breathing air are generally not adequate for process air that needs to be clean, dry and oil free.
The primary contaminants for air used in industrial applications are Particles, Water, Oil, and in some cases – Microorganisms and Gases. ISO 8573-1 Purity Classes provide acceptable limits for a variety of applications ranging from clean, dry, and oil-free to general purpose air used for industrial tools and pneumatic-fluid power-operated machines. ISO 8573-1:2010 Purity Classes are aligned with currently available purification filters. Compressed air testing is the only way to know if your purification filters are performing as expected.
ISO 8573-1 provides definitions that are key to understanding why breathing air specifications may be inappropriate for industrial / manufacturing compressed air quality.
Common Breathing Air Specifications and Typical ISO Purity Classes
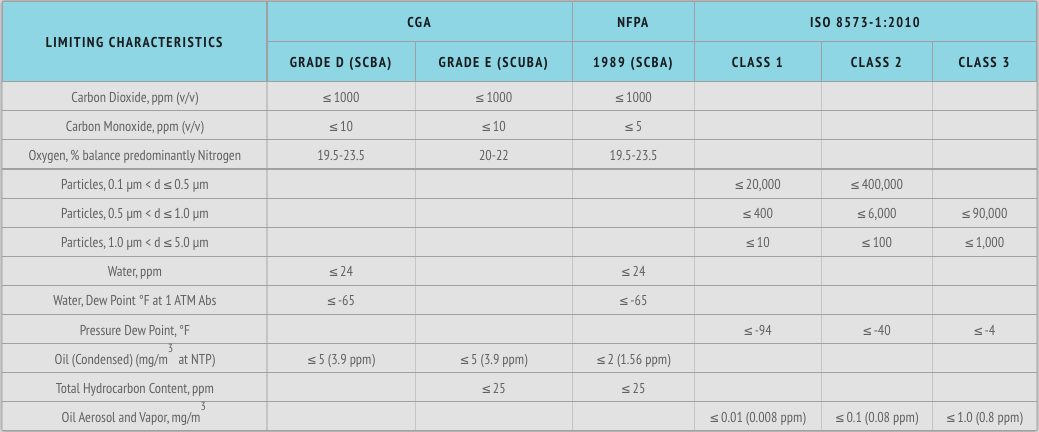
Some breathing air specifications (but not all) include Total Volatile Hydrocarbons (sometime called gaseous hydrocarbons) and all include Condensed Hydrocarbons (sometimes called oil mist or oil aerosol). The limit for Condensed Hydrocarbons or oil aerosol in breathing air ranges from 2 mg/m3 to 5 mg/m3 (apx 2 ppm – 4 ppm).
The typical laboratory analysis for Total Volatile Hydrocarbons for breathing air is an analysis for hydrocarbons composed of one to ten carbon atoms (C1 – C10). Breathing air specifications typically allow 25 ppm or do not include a limit at all whereas ISO 8573-1 Class 1 for oil which includes the combined total of both Oil Aerosol and Oil Vapor allows no more than 0.008 ppm (0.01 mg/m3) for Class 1 – the most frequently requested Purity Class for oil in critical manufacturing applications. The analysis of Oil Vapor per ISO 8573-5 calls for the analysis of hydrocarbons composed of six of more carbon atoms (typically a range of C6 – C50).
If you don’t find the air quality limits your company needs in ISO 8573-1, contact us with your custom specifications. We will be glad to provide a quote for analysis.
Why Choose Trace Analytics?
Accredited
Our A2LA accredited laboratory is an unbiased third-party laboratory with no financial interest in other compressed air products. The extensive quality control procedures at Trace’s lab ensures that you will receive quality data. Trace is an A2LA accredited laboratory complying with ISO 17025, certificate #0322-01.
Easy to Use Kits
The AirCheck✓ Kit™ is designed for use with compressed breathing air specifications requiring a dry dew point, such as ISO 8573, NFPA 1989, CGA, ISPE, and more. Other specifications are available. These kits are typically used by food, packaging, medical device, pharmaceutical, nuclear, scuba divers, fire departments and industrial breathing air facilities to test compressed air to meet their requirements.
Satisfaction
Compressed air has been our specialty since 1989 — our AirCheck✓™ Team of Experts has decades of experience in the specific field of compressed air and environmental analyses, testing, and troubleshooting. Excellent customer service, and the desire to provide a more efficient and accurate testing experience, have been the backbone of our company since 1989 — and still hold true today. We pride ourselves on the level of customer service we provide — it is the core of our relationship with all of our customers.
Industries We Serve

Testing Made SimpleAccurateEasy
Trace Analytics’ laboratory is accredited by the American Association for Laboratory Accreditation. We use state-of-the-art lab equipment that allows us to analyze hundreds of compressed air and environmental samples daily. The result is consistency, accuracy, precision, and rapid turnaround. Trace is an A2LA accredited laboratory complying with ISO 17025, certificate #0322-01.




