The Dangers of Carbon Monoxide
The dangers of Carbon Monoxide (CO) are well documented. According to the Center for Disease Control (CDC), each year more than 400 people die and 20,000 people visit the emergency room due to unintentional CO poisoning.
Compressed breathing air is not immune from this danger.
One summer, 12 students in the UK were hospitalized for carbon monoxide poisoning after taking a scuba diving class. Unfortunately some of these situations turn deadly, as in the case of a Calgary woman who died while diving off of the coast of Mexico in 2012.
Carbon Monoxide is dangerous because it competes with oxygen for binding sites in hemoglobin cells found in your blood. In high enough concentrations it can kill in less than an hour. While the effects of exposure to lower levels of Carbon Monoxide are not quite well understood, it is believed that long term exposure can cause serious, sometimes permanent damage to both the brain and heart. (Greiner 1996)
Fire departments and scuba shops must be particularly aware of the dangers that CO presents, both because strenuous activity will cause a person to take in larger amounts of contaminated air, and because of the potentially tragic consequences of CO symptoms such as disorientation and loss of consciousness in the already dangerous situations faced by firefighters and divers.
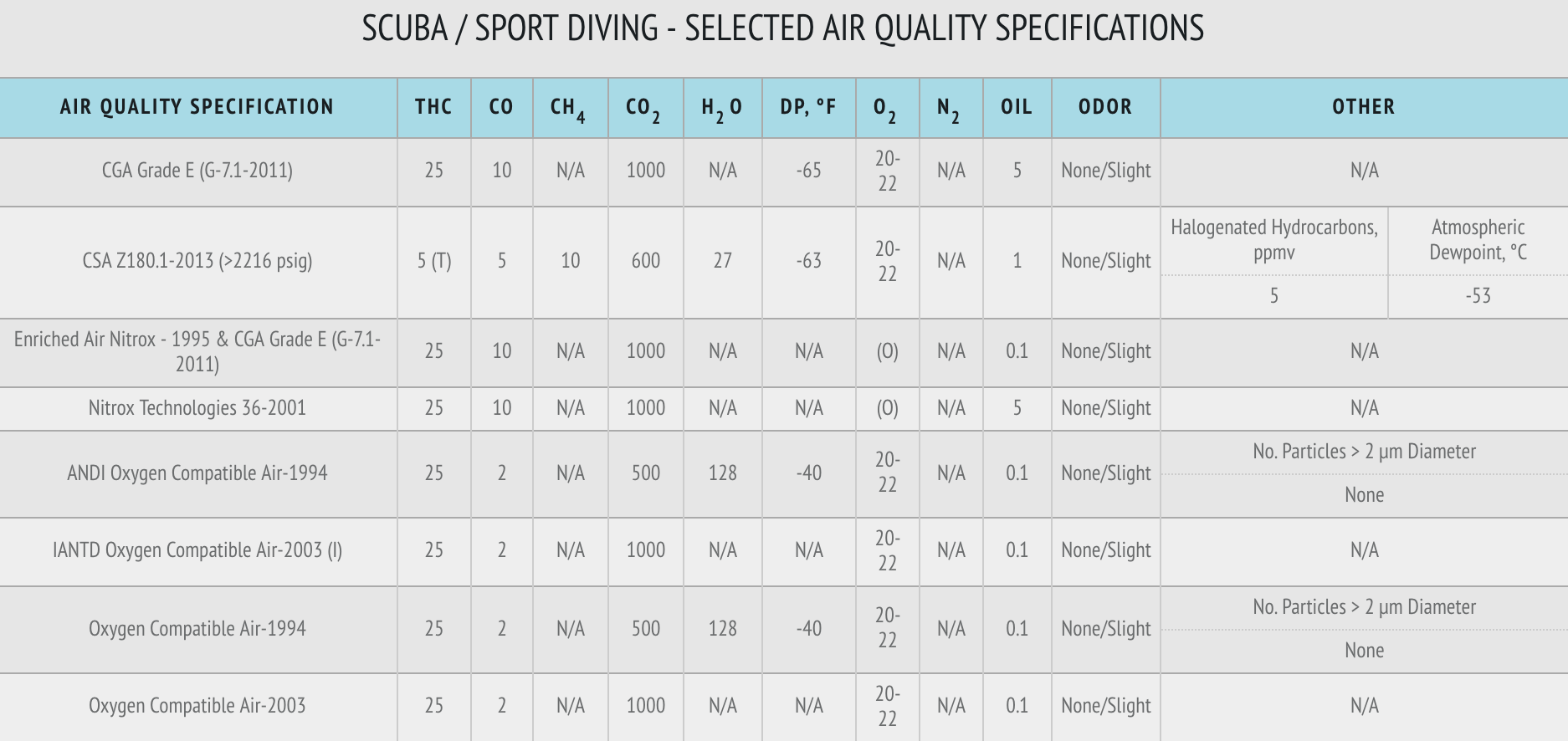
Breathing Air Specifications
What constitutes non-compliance for CO varies depending on what specification your air is being tested to. The NFPA 1989 specification, the most common specification for fire departments, sets a limit at 5 ppm, while other breathing air specifications go as high as 20.
Sources of Carbon Monoxide Contamination
Many of the sources of contamination discussed in our blog about Carbon Dioxide can also be sources of CO contamination. Like CO2, CO in compressed air mostly comes from either the compressor itself or from the air your compressor takes in. CO is a product of incomplete combustion, and some compressors have oil based lubricants that can heat up and produce Carbon Monoxide. In this instance, your results may also show elevated levels of Hydrocarbons. Ironically, if your compressor has a charcoal filter, your filter can oxidize if the compressor runs too hot, resulting in the filter contributing CO to your breathing air rather than preventing it.
Keeping your compressor well maintained can reduce these types of failures.
The most common reason for CO contamination, however, is the compressor’s intake air. While your compressor does have filters designed to remove contaminants, including CO, these filters must be used properly and changed regularly to remain effective. You can reduce this risk by ensuring your intake is in a well ventilated area and away from potential sources of Carbon Monoxide. Sending in an ambient air sample along with the sample from your compressor can help tell you if your intake air is the source of your contamination. If you know or suspect that your intake air may be the source of your contamination, try moving the intake further away from sources of carbon monoxide, or, if that’s not possible, be mindful of when your compressor is running.
Carbon Monoxide in breathing air is dangerous, but it can be avoided by being proactive in the maintenance of your compressor and by being aware of the air your compressor is taking in. By staying ahead of these problems, you can avoid the ramifications that comes with an out-of-specification air sample and, more importantly, the potentially tragic consequences of Carbon Monoxide poisoning.
References:
“Twelve pupils taken ill with suspected carbon monoxide poisoning during scuba diving lesson.” ITV News, 29 June 2017, www.itv.com/news/granada/2017-06-29/twelve-pupils-taken-ill-with-suspected-carbon-monoxide-poisoning-during-scuba-diving-lesson/.
Stewart, Nadia. “Scuba diver sues diver training organization over wife’s death in Mexico.” CBCnews, CBC/Radio Canada, 17 Dec. 2013, www.cbc.ca/news/canada/calgary/husband-of-ronda-cross-sues-scuba-training-organization-over-death-1.2466857.
Greiner, Thomas. “Carbon Monoxide Poisoning: Health Effects (AEN-166).” Department of Agricultural and Biosystems Engineering, July 1996, www.abe.iastate.edu/extension-and-outreach/carbon-monoxide-poisoning-health-effects-aen-166/.










